For my first chemistry post in a long time, I prepared 2-iodobenzoic acid from anthranilic acid using the Sandmeyer reaction. In the first step, the diazonium salt is formed by reaction of nitrous acid with anthranilic acid (nitrous acid is formed in situ from sodium nitrite and HCl). In the second step, potassium iodide is added, which allows the iodine to substitute for the diazonium ion, liberating nitrogen gas.
(yes, I know it’s not technically a Sandmeyer reaction since it doesn’t use a copper salt, but what else would you call it?)
Materials
- 1.00 g anthranilic acid
- 0.53 g sodium nitrite
- 1.23 g potassium iodide
Procedure
To a 50 mL Erlenmeyer flask, 1.00 g of anthranilic acid was added, along with 8 mL of water. 2.2 mL of 10 M HCl was then added and the flask was swirled until the anthranilic acid was fully dissolved.
Solutions of 0.53 g of sodium nitrite in 4 mL of water and a solution of 1.23 g of potassium iodide in 2 mL of water were prepared in separate containers.

The flask containing the anthranilic acid was placed in an ice bath on a magnetic stirrer, and vigorous stirring was initiated.

Once the contents of the flask had cooled, the sodium nitrite solution was added dropwise using a pipette. The reaction mixture was allowed to stir for 5 minutes.
The potassium iodide solution was then added to the reaction mixture all at once. Some effervescence was observed and a reddish-brown precipitate immediately formed.
 The reaction mixture was removed from the ice bath and allowed to stand at room temperature for 5 minutes. It was then placed in a ~42ºC water bath with moderate stirring until most of the brown precipitate had redissolved and some crystals of elemental iodine were observed floating on top of the solution.
The reaction mixture was removed from the ice bath and allowed to stand at room temperature for 5 minutes. It was then placed in a ~42ºC water bath with moderate stirring until most of the brown precipitate had redissolved and some crystals of elemental iodine were observed floating on top of the solution.
 The reaction flask was then placed directly on a hot plate and heated to ~95ºC. This temperature was maintained for 12 minutes. Temperature control is critical here: overheating will cause the iodine present to vaporize and salicylic acid will form instead of 2-iodobenzoic acid. After heating is finished, a pinch of sodium metabisulfite is added to reduce any excess elemental iodine present. The solution will still be slightly yellow, and there should be a dense, tan precipitate of crude 2-iodobenzoic acid.
The reaction flask was then placed directly on a hot plate and heated to ~95ºC. This temperature was maintained for 12 minutes. Temperature control is critical here: overheating will cause the iodine present to vaporize and salicylic acid will form instead of 2-iodobenzoic acid. After heating is finished, a pinch of sodium metabisulfite is added to reduce any excess elemental iodine present. The solution will still be slightly yellow, and there should be a dense, tan precipitate of crude 2-iodobenzoic acid.
This picture shows a successful reaction on the right, and a failed reaction on the left (most of the precipitate seen is white crystals of salicylic acid formed by reaction of the diazonium salt with water at elevated temperatures).
The crude product was collected by vacuum filtration and scraped into a 50 mL beaker while still slightly damp.

The product was dissolved in 5 mL of 95% ethanol. To this, 15 mL of hot (~50ºC) water was added, and the solution was allowed to cool to room temperature before being placed in an ice bath. Small tan, needle shaped crystals of 2-iodobenzoic acid crystallized out. The crystals were collected by vacuum filtration, rinsed three times with ice-cold water, left on the filter until mostly dry, and placed in a desiccator over calcium chloride for two days.
Characterization
First, a qualitative test was performed to detect the presence of salicylic acid in the product. A few mg of product was dissolved in a few mL of sodium bicarbonate solution. The same was done with some of the failed reaction product (shown above) and a control (pure salicylic acid). A drop of dilute iron(III) sulfate solution was added to each solution, and the solutions were re-acidified using 10 M HCl. The tube containing the brown product (left) remained colorless, while the other two immediately turned dark purple.
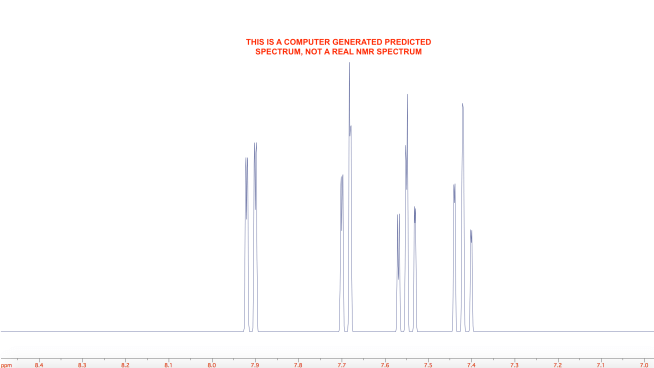
A sample of the product was analyzed by 1H-NMR. I couldn’t find a literature reference for the exact compound, but compared to a predicted spectrum, it looks very clean. The full spectrum is shown below. The rightmost peak is likely water (probably from the chloroform-d which was used as the solvent). The other peaks outside of the aromatic region are probably an impurity. The faint “lump” seen around 11.5 ppm is probably the carboxylic acid proton.
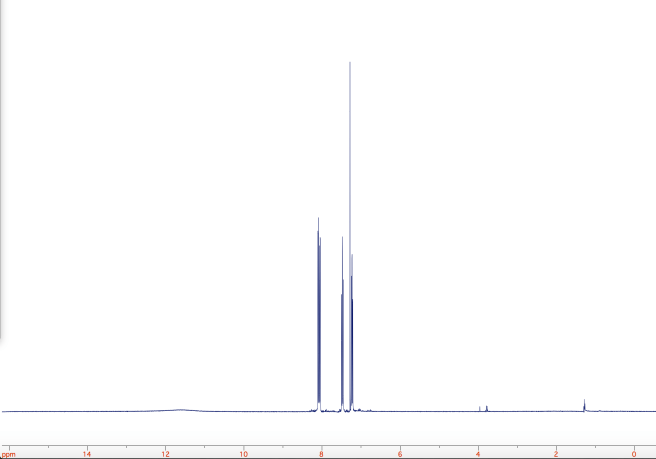
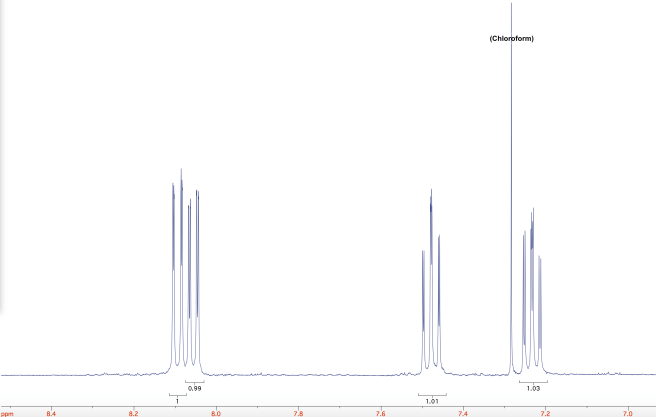
Here is a closer look at the aromatic region, where all the action is. As you can see, the integrals came out very nice!
This was the predicted spectrum:
Discussion
Yield was not stellar: 41% (0.75 g). This is probably mostly due to some spillage during the workup. More care could be taken during the recrystallization too, as there was likely a substantial amount of product remaining in solution since I filtered hastily before it was completely cooled. I am relatively satisfied with the results though, especially with the apparent purity of the product.
This product will be used to try making 2-iodoxybenzoic acid (IBX) on a small scale. IBX is a useful oxidizer that will selectively oxidize primary alcohols to aldehydes under mild conditions. I am hoping that it would be possible to recycle the oxidant somehow, since the synthesis of it is four steps long (phthalic anhydride -> phthalimide -> anthranilic acid-> 2-iodobenzoic acid-> IBX). If IBX can be recycled by re-oxidizing it, this would allow the reduced form to be extracted from reaction mixtures and “re-activated.” I am hoping that it is possible: I can’t find any information on it since it isn’t something that professional chemists would bother with. It would be a valuable procedure for the amateur though.
Relevant Links
Preparation of Anthranilic Acid
The Sandmeyer Reaction: 2-iodobenzoic acid
(Originally from: K. L. Williamson, Macroscale and Microscale Organic Experiments, 2nd Ed. 1994, Houghton Mifflin, Boston p 558, Revised 10/31/98)
Very Interesting.
I might eventually try this myself as a step before trying to prepare some Benzyl cyanide.
I found a paper about it
Oxidative decarboxylation of a-amino acids to nitriles using o-iodoxybenzoic acid in aqueous ammonia
Click to access 8303×0607.pdf
Keep the experiments coming, very informative and fun to see.
LikeLike